What is periodicity with reference to the periodic table?« Back to Questions List
|
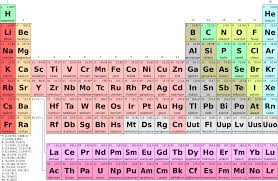
The modern periodic table is one interesting classification of chemical elements. It comprises 112 named elements as well as many unnamed ones too. The elements are arranged according to their increasing atomic number. By doing so, scientists could observe what is known as periodicity. It refers to the periodic or repetitive pattern in the chemical and physical properties of elements. Nanotechnology: Technology of the future
The four important periodic properties are ionization energy, atomic radius, electro negativity and electron affinity. Ionization energy is the energy required to remove an electron from a gaseous atom or an ion. The ability of an atom to form a chemical bond is called electro negativity and the ability of an atom to accept an electron is called electron affinity. Atomic radius is half the distance between the centers of two atoms that are touching each other.
5 Great ideas:Inventions and discoveries of great impact
Moving left to right across a row (period) of the periodic table, one may find that the ionization energy increases, electro negativity increases and atomic radius decreases. On the other hand while moving top to bottom along a column (group) of the periodic table, the ionization energy decreases, electro negativity decreases and atomic radius increases. Electron affinity normally increases across a period and sometimes decreases down a group.
Why does solid Iodine crystals on heating turn into vapor instead of melting?
|