What are the various types of chemical substances?« Back to Questions List
|
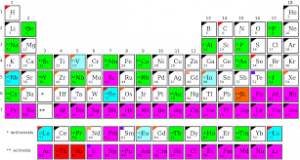
Elements, compounds and mixtures are how chemical substances are classified. Elements consist of only one kind of atom. They generally consist of either single atom or molecules (with two or more atoms) of same kind. For example, atoms of element Helium which are unreactive, exist as single atom. The atoms neither react with similar atoms, nor with other atoms of other elements. The element Oxygen (O2) consists of a two atoms of oxygen (one molecule) while nitrogen consists of 7 atoms of same kind. Some elements like Helium, Platinum are unreactive while others like sodium, magnesium are very reactive. Compounds are two or more elements joined together. Atoms of different elements join together by chemical reactions to form chemical bonds between them. The chemical bonds are nothing but a strong force that holds the atoms together. A chemical reaction alone can separate its constituent elements. The properties of compounds are quite different from the properties of the elements that make up the compound. Many things that we see are mixtures – air, rocks, sea etc. Mixtures consist of two or more elements with or without compounds physically mixed. The molecules of different elements and compounds get simply mixed without any chemical bonds. Processes like filtration, evaporation, distillation etc can easily separate the mixture.
Mixtures are of two kinds – homogeneous and heterogeneous mixtures.
Goods and Service Tax (GST) and IndiaMonsoon-Basics That Should Be Known |