How do scientists find out the age of old organic objects?« Back to Questions List
|
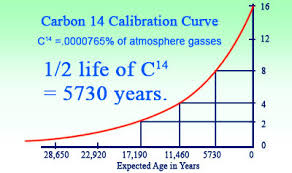
Scientists calculate the age of very old organic objects or that of human remains with a technique called radiocarbon dating. Radiocarbon is not the normal carbon atom which contains 6 protons and 6 neutrons but carbon atom with 6 protons and 8 neutrons. Hence it is called carbon-14 or radiocarbon. This carbon-14 is radioactive in nature which means it slowly decays into nitrogen-14 over a very long period of time. The radioactive substances have a definite half-life period. Half life period is the time required for exactly half of the substance to decay.
High energy rays like gamma rays or x rays that originate from the celestial objects enter the earth’s atmosphere. They react with the atoms of the atmosphere and create energetic neutrons. The collision of neutrons causes generation of Carbon-14 atoms from the nitrogen atoms (7 protons and 7 neutrons) present in the atmosphere. It gets converted into carbon-14 along with a hydrogen atom.
The carbon-14 thus created combines with oxygen to form carbon dioxide. This gas is taken in by the plants and further these plants get eaten up by animals and human. Thus carbon-14 finds its way into the living things. It starts decaying slowly and they get replaced by new carbon atoms. The measurement of the remaining proportion of carbon-14 in organic matter thus gives an estimate of its age. The half life of radiocarbon is 5700 years. It has been only reliable for dating objects up to about 60000 years old.
What are raincoats made of?Who are called primates? |