How do glow in the dark toys work?« Back to Questions List
|
We all might have admired ‘glow in the dark’ toys in our childhood days. Let us learn what makes them appear bright when there is no light at all. These toys contain a substance called phosphors (not to be confused with the chemical element phosphorus). Any material can emit light in two ways either by incandescence property (by absorbing heat) or luminescence property (by absorbing light). Phosphor is a substance that gives out visible light when energized meaning when light is there, they get charged up. They can then provide light either for minutes or hours later. These glowing phenomena could be of two types - Phosphorescence and chemiluminescence.
Phosphorescence is all about how the molecules in phosphorescent materials react to light. Chemiluminescent objects use chemical energy to produce light.
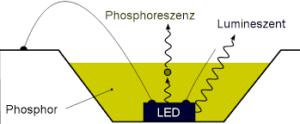
When light falls on the object, photons (packets of energy) interact with the atoms of the object. Few electrons in the object absorb the photons colliding with them and move to higher energy state for a temporary time period. This interaction of electrons and photons end, when light goes off. In the absence of light, the temperature of the object decreases and eventually the electrons return to their ground state. When they do so, they release photons (emit light). Zinc sulfide and Strontium Aluminate are two phosphors that are used in making glow in the dark toys.
A chemiluminescent object like a light stick or glow stick uses energy from a chemical reaction to emit light. Generally these sticks contain hydrogen peroxide solution, a solution containing phenyl oxalate ester and a fluorescent dye. These two solutions are kept in separate chambers. When we bend the light stick, it causes them to mix and this reaction causes electron excitation and release of photons (emission of light). Depending on the compounds used, the chemical reaction may take few minutes or go on for few hours. How different are whales from fish?How do birds prepare themselves for migration? |