Coolants and Radiators: Points to be remembered
A coolant is a fluid being used to prevent devices from overheating. The coolant flows around or through a device absorbing the heat generated by the device. The heat thus absorbed is transferred to other devices that use or dissipate heat. The term coolant is frequently used in automotive industry.
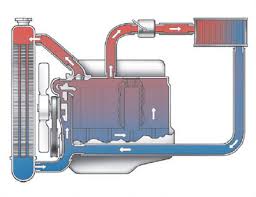
Radiators are used either for heating or cooling the coolant passing through it. Their function is transferring thermal energy from one medium to another. For radiators, the name itself is confusing as they transfer heat via convection (not by thermal radiation) contrary to what the name indicates. Radiators find their application in automobiles, electronic equipments and buildings. It acts as a source of heat for the surroundings.
The cooling system in a car prevents the car from a meltdown. Around 75% of energy produced by burning fuel in a vehicle is waste and unless the heat thus generated is removed, the engine would become a solid block of metal.
The car engine and radiator get heated up while the car is running since only around 25% of the energy produced by burning fuel is used for the forward movement. The rest becomes heat which is removed from system either with the help of radiator or as hot gas through the tail pipe. When the engine is more hot, a greater portion of the energy becomes available. If the radiator is filled with water, maximum heat it can withstand is 100 degrees as it is the boiling point of water. If the radiator cap is tightened, as in a pressure cooker, the pressure in the radiator goes up making the radiator more efficient. To raise the boiling point above 100 degrees, a coolant is mixed with water. This increases the boiling point and enhances the efficiency of the radiator.
During cold winters, plain water would freeze inside the engine and radiator. This has two disadvantages. The coolant cannot flow immediately when engine is started. The engine block or radiator may crack as the volume of ice is more than water. Therefore, coolant (anti-freeze) is essential to prevent freezing in regions of harsh winters.
Thus the coolant should be soluble in water. It should have a lower freezing point and higher boiling point. Two extensively used coolants are ethylene glycol and propylene glycol.









Informative. Keep it up
Basics in simple languiage. Would be great if you provide details of further references, if required.
Informative. Facts should be known while handling vehicle, but often unaware. Nice !!! Wait for more such articles of practical use.
Whatever stated is useful. Feel it could have covered some more details.
Cool !!!!! LOL